The pathways and domain specificity of Cu(i) binding to human metallothionein 1A
Literature Information
Adyn Melenbacher, Natalie C. Korkola, Martin J. Stillman
Copper is an essential element, but as a result of numerous adverse reactions, it is also a cellular toxin. Nature protects itself from these toxic reactions by binding cuprous copper to chaperones and other metalloproteins. Metallothionein has been proposed as a storage location for Cu(I) and potentially as the donor of Cu(I) to copper-dependent enzymes. We report that the addition of Cu(I) to apo recombinant human metallothionein 1a cooperatively forms a sequential series of Cu(I)–cysteinyl thiolate complexes that have specific Cu(I) : MT stoichiometries of 6 : 1, 10 : 1, and finally 13 : 1. The individual domain Cu : SCys stoichiometries were determined as Cu6S9 (for 6 : 1), Cu6S9 + Cu4S6 (for 10 : 1), and Cu6S9 + Cu7S9 (for 13 : 1) based on the number of modified free cysteines not involved in Cu(I) binding. The stoichiometries are associated with Cu–SCys cluster formation involving bridging thiols in the manner similar to the clusters formed with Cd(II) and Zn(II). The locations of these clustered species within the 20 cysteine full protein were determined from the unique speciation profiles of Cu(I) binding to the β and α domain fragments of recombinant human metallothionein 1a with 9 and 11 cysteines, respectively. Competition reactions using these domain fragments challenged Cu(I) metallation of the βα protein, allowing the sequence of cluster formation in the full protein to be determined. Relative binding constants for each Cu(I) bound are reported. The emission spectra of the Cu4S6, Cu6S9, and Cu7S9 clusters have unique λmax and phosphorescent lifetime properties. These phosphorescent data provide unambiguous supporting evidence for the presence of solvent shielded clusters reported concurrently by ESI-MS. Simulated emission spectra based on the cluster specific emission profiles matched the experimental spectra and are used to confirm that the relative concentrations seen by ESI-MS are representative of the solution. Our results suggest that the availability of a series of sequential Cu(I)–thiolate clusters provides flexibility as a means of protecting the cell from toxicity while still allowing for homeostatic control of the total copper content in the cell. This mechanism provides a dynamic and reactive method of reducing the cellular free copper concentrations.
Recommended Journals
Related Literature
IF 6.367
High-performance tungsten carbide electrocatalysts for the hydrogen evolution reactionIF 6.367
Facile room-temperature growth of nanostructured CuBi2O4 for selective electrochemical reforming and photoelectrochemical hydrogen evolution reactionsIF 6.367
A hollow neuronal carbon skeleton with ultrahigh pyridinic N content as a self-supporting potassium-ion battery anodeIF 6.367
Co9S8 integrated into nitrogen/sulfur dual-doped carbon nanofibers as an efficient oxygen bifunctional electrocatalyst for Zn–air batteriesIF 6.367
Enhanced activity of catalysts on substrates with surface protonic current in an electrical field – a reviewIF 6.222
Transition metal chemistry in synthetically viable alkaline earth complexes M(Cp)3− (M = Ca, Sr, Ba)IF 6.222
Biomimetic hydrogels designed for cartilage tissue engineeringIF 6.843
A model-based comparison of Ru and Ni catalysts for the Sabatier reactionIF 6.367
Milk exosomes with enhanced mucus penetrability for oral delivery of siRNAIF 6.843
Source Journal
Metallomics

Metallomics publishes cutting-edge investigations aimed at elucidating the identification, distribution, dynamics, role and impact of metals and metalloids in biological systems. Studies that address the “what, where, when, how and why” of these inorganic elements in cells, tissues, organisms, and various environmental niches are welcome, especially those employing multidisciplinary approaches drawn from the analytical, bioinorganic, medicinal, environmental, biophysical, cell biology, plant biology and chemical biology communities. We are particularly interested in articles that enhance our chemical and/or physical understanding of the molecular mechanisms of metal-dependent life processes, and those that probe the common space between metallomics and other ‘omics approaches to uncover new insights into biological processes. Metallomics seeks to position itself at the forefront of those advances in analytical chemistry destined to clarify the enormous complexity of biological systems. As such, we particularly welcome those papers that outline cutting-edge analytical technologies, e.g., in the development and application of powerful new imaging, spectroscopic and mass spectrometric modalities. Work that describes new insights into metal speciation, trafficking and dynamics in complex systems or as a function of microenvironment are also strongly encouraged. Studies that examine the interconnectivity of metal-dependent processes with systems level responses relevant to organismal health or disease are also strongly encouraged, for example those that probe the effect of chemical exposure on metal homeostasis or the impact of metal-based drugs on cellular processes.
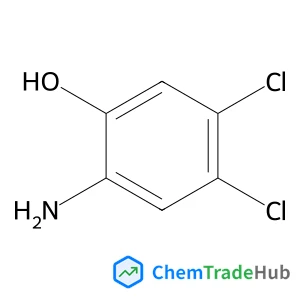
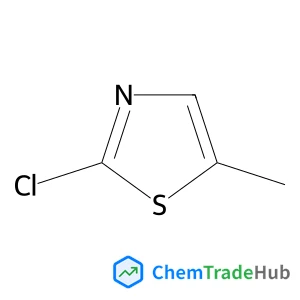
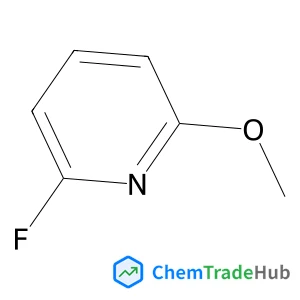
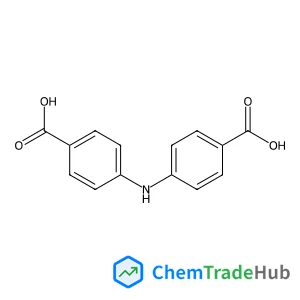
Recommended Compounds
Recommended Suppliers
 Theion GmbH
Theion GmbH DESMI GmbH
DESMI GmbH ITAS spa
ITAS spa Weibo Changsheng Nitrate Co., Ltd.
Weibo Changsheng Nitrate Co., Ltd. Dinkelberg analytics GmbH
Dinkelberg analytics GmbH Guizhou Destinate Fine Chemical Industry
Guizhou Destinate Fine Chemical Industry Dongguan Hui Xin Innovative Material Technology Co., Ltd.
Dongguan Hui Xin Innovative Material Technology Co., Ltd. Shanghai Shancuan Pump Manufacturing Co., Ltd.
Shanghai Shancuan Pump Manufacturing Co., Ltd. Verfahrenstechnik Schweitzer GmbH
Verfahrenstechnik Schweitzer GmbH Shouguang Source Beauty Food Technology Co., Ltd.
Shouguang Source Beauty Food Technology Co., Ltd.