Chiral resolution methods for racemic pharmaceuticals based on cocrystal formation
文献情報
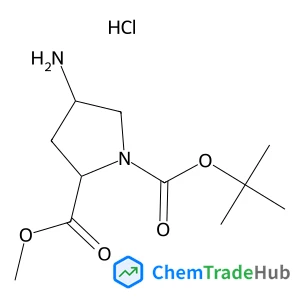
Currently, more than half of available drugs on the market are chiral, and approximately 90% of these drugs are marketed as racemates. When these racemic medications are exposed to the chiral environment of the human body, differences in their activity appear. The enantiomers of a chiral drug may potentially have distinct pharmacokinetic, metabolic, and toxicological features. As a result, regulatory requirements promote the production of enantiopure drugs to reduce the complexity of pharmacodynamics and the administration dose or to eliminate unwanted side effects, while also providing some economic savings by enabling a totally effective pharmaceutical formulation. There are a variety of procedures employed in the pharmaceutical industry for chiral separation of racemic drugs, such as developing de novo enantiomerically pure pharmaceuticals by asymmetric synthesis or chiral resolution of currently available racemic compounds via different approaches like chiral chromatography methods, diastereoisomeric salt formation, and cocrystallization-based methods. This review will focus on cocrystallization techniques such as the generation of host–guest inclusion compounds, diastereomeric cocrystal pairs, enantiospecific cocrystals, conglomerates, ionic cocrystals, and preferential enrichment of cocrystals. Overall, this review outlined the crucial importance of pharmaceutical cocrystals in chiral resolution techniques of racemic compounds.
関連文献
IF 6.367
Direct arylation polycondensation towards water/alcohol-soluble conjugated polymers as the electron transporting layers for organic solar cellsIF 6.222
Small size yet big action: a simple sulfate anion templated a discrete 78-nuclearity silver sulfur nanocluster with a multishell structureIF 6.222
Life cycle assessment of power-to-gas with biogas as the carbon sourceIF 6.367
Photoactivatable fluorophores for durable labelling of individual cellsIF 6.222
Front coverIF 6.222
Milk exosomes with enhanced mucus penetrability for oral delivery of siRNAIF 6.843
Mechanism of lignocellulose modification and enzyme disadsorption for complete biomass saccharification to maximize bioethanol yield in rapeseed stalksIF 6.367
Boronic acid liposomes for cellular delivery and content release driven by carbohydrate binding‡IF 6.222
The limits to biocatalysis: pushing the envelopeIF 6.222
掲載誌
CrystEngComm

CrystEngComm is the forum for the design and understanding of crystalline materials. We welcome studies on the investigation of molecular behaviour within crystals, control of nucleation and crystal growth, engineering of crystal structures, and construction of crystalline materials with tuneable properties and functions. We publish hypothesis-driven research into… how crystal design affects thermodynamics, phase transitional behaviours, polymorphism, morphology control, solid state reactivity (crystal-crystal solution-crystal, and gas-crystal reactions), optoelectronics, ferroelectric materials, non-linear optics, molecular and bulk magnetism, conductivity and quantum computing, catalysis, absorption and desorption, and mechanical properties. Using Techniques and methods including… Single crystal and powder X-ray, electron, and neutron diffraction, solid-state spectroscopy, spectrometry, and microscopy, modelling and data mining, and empirical, semi-empirical and ab-initio theoretical evaluations. On crystalline and solid-state materials. We particularly welcome work on MOFs, coordination polymers, nanocrystals, host-guest and multi-component molecular materials. We also accept work on peptides and liquid crystals. All papers should involve the use or development of a design or optimisation strategy. Routine structural reports or crystal morphology descriptions, even when combined with an analysis of properties or potential applications, are generally considered to be outside the scope of the journal and are unlikely to be accepted.
おすすめサプライヤー
 杭州安凯バイオمياهектив製薬有限公司
杭州安凯バイオمياهектив製薬有限公司 ジンエン(広州)新材料有限公司
ジンエン(広州)新材料有限公司 連雲港倍力達新素材有限公司
連雲港倍力達新素材有限公司 フランクGutjahrクロマトグラフ
フランクGutjahrクロマトグラフ チェムゴオーガニック株式会社
チェムゴオーガニック株式会社 MATERIAS QUÍMICAS, S. A.
MATERIAS QUÍMICAS, S. A. 盐城遠東化工有限公司
盐城遠東化工有限公司 サーモラブ科学機器株式会社 Ltd.
サーモラブ科学機器株式会社 Ltd. Interstuhl Büromöbel GmbH & Co. KG
Interstuhl Büromöbel GmbH & Co. KG 无锡開立達実業有限公司
无锡開立達実業有限公司










![221874-51-7 - 2-Methyl-2-propanyl [(3R)-2-oxo-3-piperidinyl]carbamate 221874-51-7 - 2-Methyl-2-propanyl [(3R)-2-oxo-3-piperidinyl]carbamate](/structs/221/221874-51-7-a692.webp)